Synthesis, Characterization and Applications of Silica Nanoparticles from Sugarcane Bagasse in the Removal of Tetracycline from Wastewater
| Received 22 Sep, 2025 |
Accepted 20 Jan, 2026 |
Published 21 Jan, 2026 |
Background and Objective: The persistence of tetracycline in aquatic environments poses serious ecological and health risks due to its resistance to conventional treatment methods. This study explores the potential use of sugarcane bagasse, a low-cost agricultural byproduct, as a sustainable source for synthesizing silica nanoparticles (SNPs) aimed at removing tetracycline from water. The main objective was to synthesize, characterize, and evaluate the adsorption efficiency of SNPs under varying operational conditions. Materials and Methods: Silica nanoparticles were synthesized from sugarcane bagasse via alkaline extraction and acid precipitation using the sol-gel method, a cost-effective and eco-friendly approach. The synthesized SNPs were characterized using X-ray fluorescence (XRF), X-ray diffraction (XRD), scanning electron microscopy with energy-dispersive X-ray analysis (SEM-EDX), UV-Vis spectroscopy, and Fourier-transform infrared (FTIR) spectroscopy. Adsorption experiments were conducted to evaluate the effects of pH, contact time, and adsorbent dosage on tetracycline removal. Results: The XRF analysis confirmed 96.2% silica content in the synthesized SNPs. The FTIR and UV-Vis spectra indicated the presence of Si-O-Si bonds, silanol, and siloxane groups, confirming silica formation. The XRD patterns revealed an amorphous structure, while SEM-EDX showed uniformly distributed, spherical, and porous SNPs with 60.92% silicon composition. Optimal adsorption was achieved at pH 7, 60 min contact time, and 0.8 g adsorbent dosage, demonstrating high tetracycline removal efficiency. Conclusion: The study demonstrates that sugarcane bagasse is a viable precursor for producing silica nanoparticles with strong adsorption capabilities for tetracycline removal. This approach offers a sustainable and cost-effective strategy for wastewater treatment.
| Copyright © 2026 Awodi et al. This is an open-access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. |
INTRODUCTION
Water contamination, particularly by pharmaceutical pollutants such as antibiotics, has become a pressing environmental and public health concern globally1,2. The increasing presence of antibiotics in natural water bodies, particularly tetracycline (TC), poses significant threats to ecosystems and human health3,4.
Tetracycline, widely used in both human and veterinary medicine, often enters aquatic environments through effluent from agricultural runoff, domestic sewage, and hospital waste, leading to the development of antibiotic-resistant microorganisms and disrupting aquatic biodiversity4,5. Conventional wastewater treatment processes are often inadequate for removing such contaminants, thus underscoring the need for efficient, cost-effective, and sustainable remediation technologies6,7.
Adsorption is a promising technique for the removal of antibiotics from water bodies, owing to its simplicity, ease of operation, and relatively low cost8. Agricultural waste materials, rich in silica, are being explored as potential precursors for producing adsorbent materials. Among these agricultural wastes, sugarcane bagasse presents an environmentally friendly and cost-effective source of silica9,10. With its high silica content, sugarcane bagasse offers an alternative to traditional synthetic methods of producing SNPs, reducing the need for toxic chemicals and high-energy processes, while simultaneously addressing waste disposal issues in the agro-industry11,12. To enhance the adsorption capacity and efficiency of agricultural waste-derived adsorbents, modification techniques such as nanostructuring, activation, carbonization, and grafting are employed13.
In this context, nanotechnology offers promising solutions for the removal of water pollutants, with silica nanoparticles (SNPs) gaining attention for their adsorptive properties, environmental compatibility, and versatility14-16. However, the large-scale synthesis and application of synthetic nanoparticles is hindered by issues such as aggregation, difficult separation, and potential environmental and health risks associated with their discharge17-19. To mitigate these challenges, green synthesis of silica nanoparticles is being explored. Silica nanoparticles possess desirable properties, including good physical stability and chemical inertness, making them a valuable material in various applications20. Additionally, SNPs exhibit high surface reactivity, low toxicity and straightforward chemistry further expanding their utility15-21. They are commonly employed in industrial manufacturing, packaging of ceramics, synthesis of high molecular-weight composite materials, as well as in biomedical fields such as drug delivery, cancer therapy, disease labeling, biosensing and in the food and agricultural sector22-24.
Recently, researchers have investigated the synthesis of SNPs from sugarcane bagasse. For instance, Sulaiman et al.25 and Mohd et al.26 successfully synthesized SNPs from sugarcane bagasse using an extraction-precipitation method. Additionally, Ribeiro et al.27 developed a system utilizing pristine sugarcane bagasse as an adsorbent, demonstrating its effectiveness in removing tetracycline from wastewater. Furthermore, Chen et al.28 created a magnetically modified sugarcane bagasse biochar, which showed high adsorption efficiency for tetracycline removal from wastewater. Despite attempts to synthesize SNPs from sugarcane bagasse and utilizing bagasse for adsorptive removal of tetracycline from wastewater, there is currently no study on SNPs derived from sugarcane bagasse for tetracycline removal. Given the enhanced adsorptive capacity of SNPs due to their nanoscale size, this research gap highlights the need for a study exploring the potential of SNPs from sugarcane bagasse for effective tetracycline removal from wastewater.
Therefore, this study explores the synthesis and characterization of silica nanoparticles from sugarcane bagasse using an alkaline extraction and acid precipitation process, a method known for its low cost, reduced toxicity, and lower energy requirements compared to other conventional methods. The study also investigates the adsorptive efficiency of the SNPs for the removal of tetracycline from water under various conditions, such as pH, contact time, and adsorbent dosage.
MATERIALS AND METHODS
Study area: This research took place in Markudi, Benue State, Nigeria, which is positioned at Latitude of 7.73°N and a Longitude of 5.84°E. The city’s elevation is 103 m above sea level and has a population of over 5,741,815. This research was conducted between the period of October, 2024 and September, 2025.
Materials: Sugarcane was sourced from local sellers in Markurdi, Benue State, and subsequently chopped and peeled to obtain the sugarcane bagasse. A 50 mg capsule of tetracycline was bought from a pharmacy store in Markurdi. All other chemicals and reagents used in this study, including sodium hydroxide and hydrochloric acid, were obtained from the chemical laboratory in the department of chemistry, Benue State University, Markurdi, Nigeria.
Preparation of sugarcane bagasse ash: The sugarcane bagasse was soaked in clean water overnight and washed thoroughly to remove dirt and soil. It was then dried in the sun for two days, followed by further drying in an oven at 90°C. The dried sugarcane bagasse was subsequently burnt to produce sugarcane bagasse ash (SBA). The SBA was then heated in a muffle furnace at 600°C for 3 hrs to remove the carbon content, and left overnight to cool in the furnace.
Extraction of silica from sugarcane bagasse ash (SBA): Silica was extracted from sugarcane bagasse ash using the method described by Ni’mah et al.29 with some modifications. In this method, 250 mL of 1 N NaOH was added to 10 g SBA samples and boiled in a covered 100 mL Erlenmeyer flask for 1 hr with constant stirring to dissolve the silica and produce a sodium silicate solution. The solution was then filtered through Whatman. No. 1 filter paper, and the residue was washed with boiled distilled water. The filtrate was allowed to cool to room temperature, and the pH of the solution was adjusted to 7 using 3 N HCl with constant stirring. The solution was then incubated to promote gel formation. Once the gel had formed, it was aged for 18 hrs. After ageing, the soft gel was gently broken, and the slurry was centrifuged at 4000 rpm for 5 min. The supernatant was discarded, and the gel was transferred to a beaker and dried at 80°C for 24 hrs to produce xerogels. The silica xerogels were subjected to additional washing with deionized water to effectively remove minerals and impurities from the silica. This step is more effective when performed on the dried silica (xerogel) rather than on the silica gel (aqua-gel) before drying. Finally, the silica was dried in a hot air oven at 105°C for 24 hrs.
Preparation of silica nanoparticles (SNPs): Silica nanoparticles were prepared from the extracted silica using a refluxing technique following the method of Elnazer et al.30 with some modifications. The extracted silica was refluxed with 6 M HCl at 85oC for 4 hrs and washed repeatedly with distilled water to remove any residual acid. The silica was then dissolved in 2.5 M NaOH with continuous stirring, and sulfuric acid was added until the pH reached 8, resulting in the precipitation of silica. The precipitated silica was washed repeatedly with warm distilled to remove any residual alkali and then dried in a hot air oven at 50°C for 48 hrs.
Characterization
Fourier transform infrared (FTIR) spectroscopy analysis: The FTIR spectroscopy analysis was conducted based on procedures of Seroka et al.11. In this study, a Perkin Elmer (Nicolet iS10 FT-IR) spectrophotometer was employed to identify the vibrational frequencies of functional groups in the silica nanoparticles powder. The silica powder was mixed with potassium bromide (KBR) and ground into a thin layer of matrix for analysis.
X-ray powder diffraction analysis: The crystal structure and morphology of the prepared sample were characterized following the method of Omoike et al.31,32 and Seroka et al.11 using an X-ray powder diffractometer (Rigaku Miniflex 600, Japan). The XRD measurements were taken with a step size of 0.02°/min, utilizing Cu Kα radiation filtered with Ni.
Scanning electron microscopy (SEM): The size, shape, and surface morphology of the prepared silica nanoparticles were examined using a Scanning Electron Microscope (Phenom ProX by PhenomWorld Eindhoven, Netherlands) by following procedures adopted by Elnazer et al.30 and Omoike et al.33. This analysis provided high-resolution images of the nanoparticles at 7000-8000 times magnification, allowing for a detailed characterization of their surface morphology and particle size distribution.
Brunauer-emmert-teller (BET) analysis: The surface area and pore volume of the silica nanoparticles were determined using a BET analyzer based on Hoang et al.34. This analysis involved measuring the nitrogen adsorption-desorption isotherms, which provided information on the specific surface area, pore size distribution and total pore volume of the nanoparticles.
Preparation of tetracycline stock solution: Tetracycline stock solution was prepared as described by Elnazer et al.30 with modifications. In this method, 1 g of tetracycline was dissolved in distilled water and made up to 1000 mL volume mark in a volumetric flask. The flask was then kept for serial dilution to achieve the desired concentration.
Batch adsorption experiments: Batch adsorption studies were conducted to investigate the adsorption of tetracycline (TC) onto silica nanoparticles (SNPs) under various conditions. These investigations were carried out based on the methods described by Hoang et al.34 with some modifications.
Adsorbent dosage: The effect of adsorbent dosage on TC adsorption was examined by adding different masses of SNPs (0.2, 0.4, 0.6, and 0.8 g) to 20 mL of 15 mg/L TC solution at pH 7. The mixtures were stirred in a mechanical shaker for 1 hour and then filtered. The filtrates were analyzed using UV-visible spectroscopy. The percentage removal (%R) of TC was calculated using Equation 1 below:
| (1) |
Where:
| R (%) | = | Percentage of TC removed | |
| Ci | = | Initial concentration of TC | |
| Ce | = | Concentration of TC after adsorption |
pH variations: The effect of pH on adsorption was investigated by adjusting the pH of the SNPs/TC solution from 2 to 11 using 0.1M HCl and 0.1M NaOH. The mixtures were shaken for 1 hr and then filtered. The pH with the highest adsorption efficiency was determined as the optimum adsorption pH.
Contact time: The impact of contact time on adsorption efficiency was studied at varying times (20, 40, 60, and 80 min) using 20 mL of 15 mg/L TC solution with 0.4 g of SNPs at room temperature. The mixtures in 200 mL stopper conical flasks were placed in a mechanical shaker and shaken at 130 rpm.
RESULTS AND DISCUSSIONS
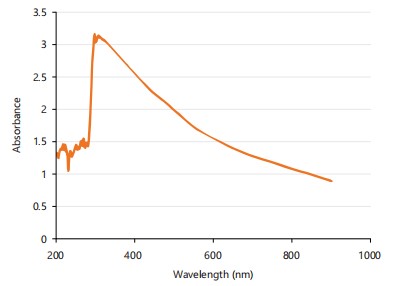
UV-visible spectroscopy analysis: UV-Vis spectroscopy is a widely used technique for characterizing the optical properties of nanoparticles, providing valuable information on their absorption and transmission characteristics35. Figure 1 shows the UV-Vis absorption spectrum of silica nanoparticles, which is a plot of the optical absorbance as a function of wavelength. The result shows that the UV-visible spectrum of SNPs recorded a maximum absorption band edge of 310 nm. These optical features are similar to those obtained in previous reports where the maximum absorption peaks were around 300-390 nm and attributed to the Si-O-Si bond, confirming the presence of silica nanoparticles35,36.
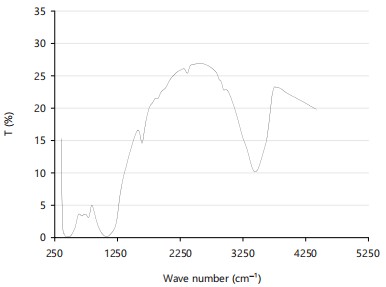
FTIR spectroscopic analysis: The FTIR spectrum of SNPs as shown in Fig. 2 and Table 1 revealed the following: 1088-1632/cm indicating Si-O-Si, and Si-O stretching vibration, 400/cm corresponding to Si-O-Si asymmetric bend, 3442/cm is assigned to Si-OH due to surface bonding to water molecules37, 2250-2750/cm corresponds to Si-H, a siloxane group, and the predominant absorbance peak at 1320/cm was due to siloxane bonds (Si-O-Si). The FTIR Spectrum has clearly shown the presence of SNPs when compared with Silica peaks reported in the literature38.
|
|
| Table 1: | FTIR absorption peaks of SNPs from SBA | |||
| Wave number (cm-1) | Functional group |
| 400 | Si-O-Si, asymmetric bend |
| 1088-1632 | Si-O-Si, SiO stretch |
| 2250-2750 | Si-H |
| 3442 | Si-OH |
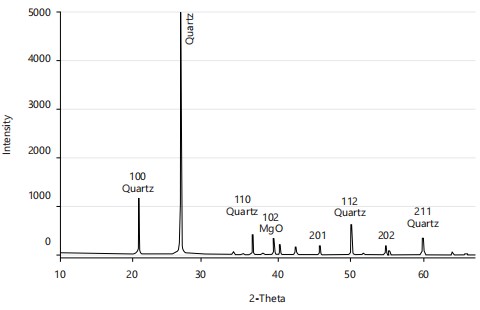
X-ray diffraction: The X-ray diffraction pattern of SNPs is displayed in Fig. 3. The XRD intense sharp peaks were observed at 2θ (2-theta) = 22.3°, 26.8°, while low peaks were observed at 2θ = 37.2°, 50.1°, 60.0° which confirms the combination of crystal and amorphous structure of silica nanoparticles. Similar results were recorded with XRD sharp peaks at 2θ = 22-30° silica nanoparticles obtained from rice husk30.
Scanning electron microscopy: The surface morphology of SNPs was analyzed using SEM and the result is presented in Fig. 4a-b. The SEM image displayed a uniform distribution of silica nanoparticles in the form of roughly spherical shapes with some degree of pore spaces. This indicates a porous microsurface structure of SNPs. This result is consistent with the findings of Elnazer et al.30 who reported similar spherical and homogenously distributed silica nanoparticles synthesized from rice husk, as observed in the SEM images.
|

|
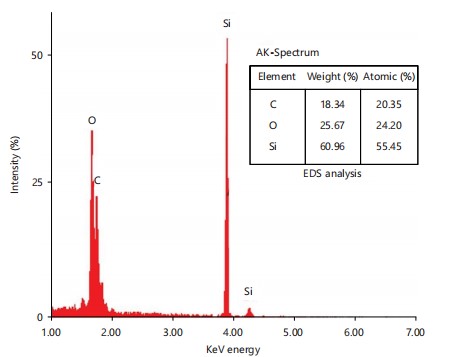
The elemental composition of the SNPs was determined by EDX, as shown in Fig. 5. The results revealed that silicon was the predominant element, constituting 60.96% w/w of the composition, followed by oxygen at 25.67% w/w and carbon at 18.34% w/w. These findings confirm that sugarcane bagasse is a rich source of silicon, which was successfully extracted and utilized in the synthesis of SNPs.
In addition, the XRF analysis as shown in Table 2 shows the SNPs composition data indicating that SiO2 content was 96.2, also confirms the high amount of the silica nanoparticle produced39.
Brunauer-emmert-teller (BET) analysis: Table 3 shows BET surface area, average pore size, and pore volume of SNPs prepared from sugarcane bagasse. The results revealed that SNPs had a characteristics surface area of 90.720 m2/g, average adsorption pore size 28.5500 A°, and total pore volume 0.152240 cm3/g. These characteristics indicate a microporous structure that is conducive to effective adsorption. The relatively high surface area and pore volume of the SNPs, although lower than some previous studies, suggest that SNPs derived from sugarcane bagasse are suitable for antibiotic removal applications, thus providing ample active sites for adsorption34. The microporous structure also enables efficient interaction between the adsorbent and target pollutants, underscoring the potential of SNPs as effective adsorbents in water treatment processes.
|
| Table 2: | XRF analysis of silica nanoparticles from SBA | |||
| Element/compound | Amount |
| SiO2 | 96.2 |
| AlO3 | - |
| Fe2O3 | 0.8 |
| T1O2 | 0.23 |
| CaO | 0.33 |
| P2O5 | 0.06 |
| K2O | 1.24 |
| MnO | 0.02 |
| MgO | - |
| Na2O | 1.1 |
| Cu | 0.01 |
| Zn | 0.001 |
| Rb | 0.01 |
| Cr | 0.01 |
| Zr | 0.02 |
| Total | 100 |
| Table 3: | BET surface area and pore characteristics of silica nanoparticles from SBA | |||
| Sample | Surface area (m2/g) | Average pore size (A°) | Pore volume (cm3/g) |
| Silica nanoparticles | 92.72±3.22 | 30.52±2.06 | 0.15224 |
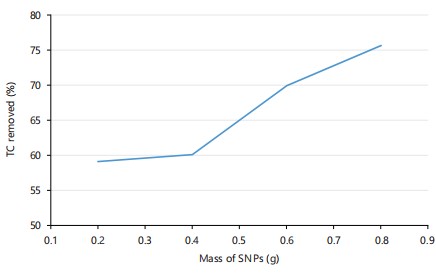
Effect of SNPs adsorbent dosage: The effectiveness of adsorption relies heavily on the availability of active sites on the adsorbent surface, making adsorbent dosage a critical factor in determining the efficiency of the adsorption process30. Figure 6 presents the effect of silica nanoparticles (SNPs) adsorbent dosage on tetracycline (TC) adsorption within an adsorbent range of 0.2-0.8 g at a pH of 7. The results showed that increasing the SNPs dosage led to an increase in TC adsorption percentage. Initially, there was a slow increase in the adsorption capacity of SNPs from 59.07 to 60.06% at a dosage range of 0.2-0.4 g. This was then followed by a significant increase in TC adsorption percentage by 16.3% occurring between 0.4 and 0.6 g SNPs dosage. Comparatively, the adsorption decreased slightly between 0.6 to 0.8 g SNPs dosage. The increase in adsorption capacity with increasing adsorbent dosage can be attributed to the availability of more adsorption sites and enhanced surface area40. As the adsorbent dosage increases, the total surface area and charge density of the adsorbent also increase, providing more binding sites for TC adsorption. This trend is consistent with previous studies, which suggest that increased adsorbent dosage enhances adsorption capacity due to increased surface area and availability of adsorption sites39-41.
|
|
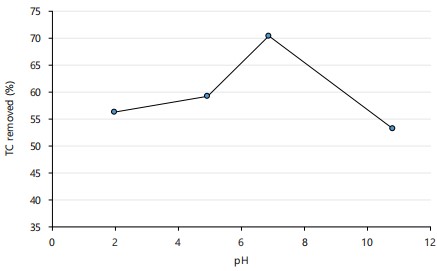
Effect of pH: Adsorption processes are greatly influenced by the pH of the medium as it determines the specific nature of adsorbates and their surface charge27. The effect of pH on tetracycline (TC) adsorption onto silica nanoparticles (SNPs) is presented in Fig. 7. The results show that the adsorption of TC increased significantly from 59.74 to 71.16 by 19.1% with an increase pH from 5 to 7. There was, however, a sharp decrease by 24.5% from pH 7 to 11. The maximum adsorption capacity of SNPs was achieved at pH 7, where TC exists in its zwitterionic form42. This suggests that TC adsorption onto SNPs is primarily driven by non-electrostatic interactions43. At pH <6.0, low adsorption of TC was observed due to repulsive forces between positively charged TC species and the SNPs surface44. Conversely, at pH >9.0, the amount of TC adsorbed/removed decreased due to silica dissolution and desorption at high pH values40. Therefore, pH 7.0 was determined as the optimal pH for TC removal using SNPs, and this value was used for further investigations.
|
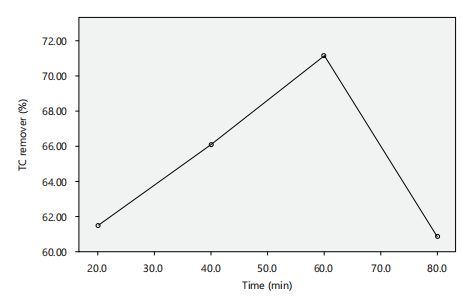
Effect of contact time on TC adsorption: The contact time between the adsorbent and adsorbate plays a significant role in determining the rate and efficiency of adsorption45. Therefore, investigating the effect of contact time on tetracycline (TC) adsorption onto silica nanoparticles (SNPs) is essential to understand the adsorption kinetics and possible optimization of the process. Figure 8 shows the effect of contact time on tetracycline adsorption at an initial concentration of 0.15 mg/L. The results revealed that there was a rapid adsorption of TC in the initial stage, with equilibrium attained within 60 min. TC removal increased sharply with increasing adsorption time from 20 to 60 min, after which the change in TC removal became insignificant. This indicates that the adsorption equilibrium was reached at 60 min, and desorption occurred thereafter30-46. Therefore, an adsorption time of 60 min was determined as optimal and used for further studies on TC removal using SNPs.
CONCLUSION
This study successfully synthesized and characterized silica nanoparticles (SNPs) from sugarcane bagasse, confirming their pure form and structure through various analytical techniques. The SNPs exhibited a maximum absorption band edge of 310 nm, characteristic functional groups, an amorphous structure, and a high BET surface area. The batch adsorption studies demonstrated optimal conditions for tetracycline adsorption, with contact times, pH, and adsorbent dosages of 60 min, 7, and 1.0 g, respectively. Based on the results, it can be concluded that the use of sugarcane bagasse as a source of SNPs demonstrated a promising and efficient adsorbent in the removal of tetracycline from wastewater, providing a sustainable solution for water treatment. Moreover, utilizing sugarcane bagasse as a source of SNPs offers environmental and economic benefits by valorizing agricultural waste. The synthesized SNPs show potential for various industrial and agricultural applications. This research demonstrates the potential of converting abundant agricultural waste into valuable nanomaterials for environmental remediation.
SIGNIFICANCE STATEMENT
This study discovered the potential of sugarcane bagasse, an abundant agricultural byproduct, as an effective precursor for synthesizing silica nanoparticles capable of removing tetracycline from water. The research highlights a sustainable and low-cost method to address the growing issue of antibiotic contamination in aquatic environments, which conventional treatments often fail to mitigate. By demonstrating high adsorption efficiency under optimized conditions, this work bridges the gap between agricultural waste utilization and environmental remediation. This study will help researchers to uncover the critical areas of nanoparticle-based adsorption mechanisms and green nanomaterial synthesis that many researchers were not able to explore. Thus, a new theory on sustainable nanotechnology for wastewater treatment may be arrived at.
REFERENCES
- Chaturvedi, P., P. Shukla, B.S. Giri, P. Chowdhary, R. Chandra, P. Gupta and A. Pandey, 2021. Prevalence and hazardous impact of pharmaceutical and personal care products and antibiotics in environment: A review on emerging contaminants. Environ. Res., 194.
- Morin-Crini, N., E. Lichtfouse, G. Liu, V. Balaram and A.R.L. Ribeiro et al., 2022. Worldwide cases of water pollution by emerging contaminants: A review. Environ. Chem. Lett., 20: 2311-2338.
- Amangelsin, Y., Y. Semenova, M. Dadar, M. Aljofan and G. Bjørklund, 2023. The impact of tetracycline pollution on the aquatic environment and removal strategies. Antibiotics, 12.
- Xu, L., H. Zhang, P. Xiong, Q. Zhu, C. Liao and G. Jiang, 2021. Occurrence, fate, and risk assessment of typical tetracycline antibiotics in the aquatic environment: A review. Sci. Total Environ., 753.
- Gomes, M.P., 2024. The convergence of antibiotic contamination, resistance, and climate dynamics in freshwater ecosystems. Water, 16.
- Shamshad, J. and Rashid Ur Rehman, 2025. Innovative approaches to sustainable wastewater treatment: A comprehensive exploration of conventional and emerging technologies. Environ. Sci.: Adv., 4: 189-222.
- Shah, A.I., M.U.D. Dar, R.A. Bhat, J.P. Singh, K. Singh and S.A. Bhat, 2020. Prospectives and challenges of wastewater treatment technologies to combat contaminants of emerging concerns. Ecol. Eng., 152.
- Narwal, N., D. Katyal and N. Kataria, 2023. Biomaterials and Eco-Friendly Materials for the Sequestration of Pharmaceuticals from Contaminated Water. In: Pharmaceuticals in Aquatic Environments, Garg, V.K., A. Pandey, N. Kataria and C. Faggio (Eds.), CRC Press, Boca Raton, Florida, ISBN: 9781003436607, pp: 39-64.
- September, L.A., N. Kheswa, N.S. Seroka and L. Khotseng, 2023. Green synthesis of silica and silicon from agricultural residue sugarcane bagasse ash-a mini review. RSC Adv., 13: 1370-1380.
- Kusuma, H.S., D. Permatasari, W. Khoirul Umar and S.K. Sharma, 2024. Sugarcane bagasse as an environmentally friendly composite material to face the sustainable development era. Biomass Convers. Biorefin., 14: 26693-26706.
- Seroka, N.S., R.T. Taziwa and L. Khotseng, 2022. Extraction and synthesis of silicon nanoparticles (SiNPs) from sugarcane bagasse ash: A mini-review. Appl. Sci., 12.
- Wu, S.H., C.Y. Moua and H.P. Lin, 2013. Synthesis of mesoporous silica nanoparticles. Chem. Soc. Rev., 42: 3862-3875.
- Mo, J., Q. Yang, N. Zhang, W. Zhang, Y. Zheng and Z. Zhang, 2018. A review on agro-industrial waste (AIW) derived adsorbents for water and wastewater treatment. J. Environ. Manage., 227: 395-405.
- Baruah, S., M.N. Khan and J. Dutta, 2016. Perspectives and applications of nanotechnology in water treatment. Environ. Chem. Lett., 14: 1-14.
- Li, H., X. Chen, D. Shen, F. Wu, R. Pleixats and J. Pan, 2021. Functionalized silica nanoparticles: Classification, synthetic approaches and recent advances in adsorption applications. Nanoscale, 13: 15998-16016.
- Akhayere, E., D. Kavaz and A. Vaseashta, 2022. Efficacy studies of silica nanoparticles synthesized using agricultural waste for mitigating waterborne contaminants. Appl. Sci., 12.
- Shrestha, S., B. Wang and P. Dutta, 2020. Nanoparticle processing: Understanding and controlling aggregation. Adv. Colloid Interface Sci., 279.
- Jamuna, B.A. and R.V. Ravishankar, 2014. Environmental Risk, Human Health, and Toxic Effects of Nanoparticles. In: Nanomaterials for Environmental Protection, Kharisov, B.I., O.V. Kharissova and H.V.R. Dias (Eds.), Wiley, New Jersey, USA, ISBN: 9781118845530.
- Omoike, B.A., M.A. Abubakar, C. Imoisi and S.J. Iduma, 2025. Application of nanotechnology in building construction using carbon nanotubes: A review. Singapore J. Sci. Res., 15: 18-29.
- Buzea, C. and I. Pacheco, 2017. Nanomaterial and Nanoparticle: Origin and Activity. In: Nanoscience and Plant-Soil Systems, Ghorbanpour, M., K. Manika and A. Varma (Eds.), Springer, Cham, Switzerland, ISBN: 978-3-319-46835-8, pp: 71-112.
- Omoike, B.A., F.E. Okieimen and C. Imoisi, 2024. Design and optimization of eco-friendly biocomposite films for packaging applications using response surface methodology. Tanzania J. Sci., 50: 961-974.
- Prabha, S., D. Durgalakshmi, S. Rajendran and E. Lichtfouse, 2021. Plant-derived silica nanoparticles and composites for biosensors, bioimaging, drug delivery and supercapacitors: A review. Environ. Chem. Lett., 19: 1667-1691.
- Videira-Quintela, D., O. Martin and G. Montalvo, 2021. Emerging opportunities of silica-based materials within the food industry. Microchem. J., 167.
- Omoike, B.A., F.E. Okieimen and C. Imoisi, 2024. Water vapour transport properties of cassava starch/poly(vinyl alcohol) films via experimental and Fickian methods. Trends Appl. Sci. Res., 19: 225-232.
- Sulaiman, M., N.I.D. Iya and M. Aliyu, 2024. Synthesis of silica nanoparticles from sugarcane waste: Precipitation-based size control and characterization. FUDMA J. Sci., 8: 222-227.
- Mohd, N.K., N.N.A.N. Wee and A.A. Azmi, 2017. Green synthesis of silica nanoparticles using sugarcane bagasse. AIP Conf. Proc., 1885.
- Ribeiro, A.V.F.N., P. da Cruz Cosmo, M. de Godoi Pereira, B.M. Dalfior and G. dos Santos Gonçalves et al., 2014. Use of sugarcane bagasse for adsorption of tetracycline in aqueous medium. Indian J. Appl. Res., 4: 10-14.
- Chen, W., M. Shen and G. Li, 2019. Highly-efficient adsorptive removal of tetracycline using magnetic sugarcane bagasse biochar modified by lanthanum. Nat. Environ. Pollut. Technol., 18: 639-643.
- Ni'mah, Y.L., Z. Hidayatul Muhaiminah and Suprapto, 2023. Synthesis of silica nanoparticles from sugarcane bagasse by sol-gel method. AIP Conf. Proc., 2540.
- Elnazer, W.Y., T.E. Farrag, M.S. Beheary and R.A. Mansour, 2024. Efficient adsorption of amoxicillin onto silica nanoparticles synthesized from rice husks. Desalin. Water Treat., 317.
- Omoike, B.A., F.E. Okieimen, C. Imoisi and M.A. Abubakar, 2024. Characterization and evaluation of properties of cassava starch/poly(vinyl alcohol) films for food and pharmaceutical packaging applications. Singapore J. Sci. Res., 14: 34-42.
- Omoike, B.A., F.E. Okieimen and C. Imoisi, 2026. Developing composite films from carboxymethyl starch, polyvinyl alcohol, and kaolin for sustainable packaging applications. Foods Raw Mater., 14: 344-356.
- >Omoike, B.A., F.E. Okieimen and C. Imoisi, 2025. Development of lemongrass oil-based starch/PVA/kaolin films: Antimicrobial properties and biodegradability. Sci. Int., 13: 1-12.
- Hoang, L.P., T.M.P. Nguyen, H.T. Van, M. Yılmaz and T.K. Hoang et al., 2022. Removal of tetracycline from aqueous solution using composite adsorbent of ZnAl layered double hydroxide and bagasse biochar. Environ. Technol. Innovation, 28.
- Biradar, A.I., P.D. Sarvalkar, S.B. Teli, C.A. Pawar, P.S. Patil and N.R. Prasad, 2021. Photocatalytic degradation of dyes using one-step synthesized silica nanoparticles. Mater. Today: Proc., 43: 2832-2838.
- Yang, M., W. Jiang, Z. Pan and H. Zhou, 2015. Synthesis, characterization and SOD-like activity of histidine immobilized silica nanoparticles. J. Inorg. Organomet. Polym. Mater., 25: 1289-1297.
- Feifel, S.C. and F. Lisdat, 2011. Silica nanoparticles for the layer-by-layer assembly of fully electro-active cytochrome c multilayers. J. Nanobiotechnol., 9.
- Heidari, F., S.M. Jafari, A.M. Ziaiifar and N. Anton, 2023. Surface modification of silica nanoparticles by chitosan for stabilization of water-in-oil Pickering emulsions. Carbohydr. Polym. Technol. Appl., 6.
- Seroka, N.S., R. Taziwa and L. Khotseng, 2022. Green synthesis of crystalline silica from sugarcane bagasse ash: Physico-chemical properties. Nanomaterials, 12.
- Hoang, L.P., T.M.P. Nguyen, H.T. Van, T.K.D. Hoang, X.H. Vu, T.V. Nguyen and N.X. Ca, 2020. Cr(VI) removal from aqueous solution using a magnetite snail shell. Water Air Soil Pollut., 231.
- Nasiri, A., N. Golestani, S. Rajabi and M. Hashemi, 2024. Facile and green synthesis of recyclable, environmentally friendly, chemically stable, and cost-effective magnetic nanohybrid adsorbent for tetracycline adsorption. Heliyon, 10.
- Hashemikia, S., N. Hemmatinejad, E. Ahmadi and M. Montazer, 2015. Optimization of tetracycline hydrochloride adsorption on amino modified SBA-15 using response surface methodology. J. Colloid Interface Sci., 443: 105-114.
- Brigante, M. and M. Avena, 2016. Biotemplated synthesis of mesoporous silica for doxycycline removal. Effect of pH, temperature, ionic strength and Ca2+ concentration on the adsorption behaviour. Microporous Mesoporous Mater., 225: 534-542.
- Qi, N., P. Wang, C. Wang and Y. Ao, 2018. Effect of a typical antibiotic (tetracycline) on the aggregation of TiO2 nanoparticles in an aquatic environment. J. Hazard. Mater., 341: 187-197.
- Aragaw, T.A. and A.N. Alene, 2022. A comparative study of acidic, basic, and reactive dyes adsorption from aqueous solution onto kaolin adsorbent: Effect of operating parameters, isotherms, kinetics, and thermodynamics. Emerging Contam., 8: 59-74.
- Khatibi, A.D., A.H. Mahvi, N. Mengelizadeh and D. Balarak, 2021. Adsorption-desorption of tetracycline onto molecularly imprinted polymer: Isotherm, kinetics, and thermodynamics studies. Desalin. Water Treat., 230: 240-251.
How to Cite this paper?
APA-7 Style
Awodi,
G.O., Omoike,
B.A., Oloche,
A.J., Imoisi,
C. (2026). Synthesis, Characterization and Applications of Silica Nanoparticles from Sugarcane Bagasse in the Removal of Tetracycline from Wastewater. International Journal of Biological Chemistry, 20(1), 1-12. https://doi.org/10.3923/ijbc.2026.01.12
ACS Style
Awodi,
G.O.; Omoike,
B.A.; Oloche,
A.J.; Imoisi,
C. Synthesis, Characterization and Applications of Silica Nanoparticles from Sugarcane Bagasse in the Removal of Tetracycline from Wastewater. Int. J. Biol. Chem 2026, 20, 1-12. https://doi.org/10.3923/ijbc.2026.01.12
AMA Style
Awodi
GO, Omoike
BA, Oloche
AJ, Imoisi
C. Synthesis, Characterization and Applications of Silica Nanoparticles from Sugarcane Bagasse in the Removal of Tetracycline from Wastewater. International Journal of Biological Chemistry. 2026; 20(1): 1-12. https://doi.org/10.3923/ijbc.2026.01.12
Chicago/Turabian Style
Awodi, Godwin, Owoicho, Bright Ayemwenre Omoike, Akpode Jacob Oloche, and Chinyere Imoisi.
2026. "Synthesis, Characterization and Applications of Silica Nanoparticles from Sugarcane Bagasse in the Removal of Tetracycline from Wastewater" International Journal of Biological Chemistry 20, no. 1: 1-12. https://doi.org/10.3923/ijbc.2026.01.12

This work is licensed under a Creative Commons Attribution 4.0 International License.



